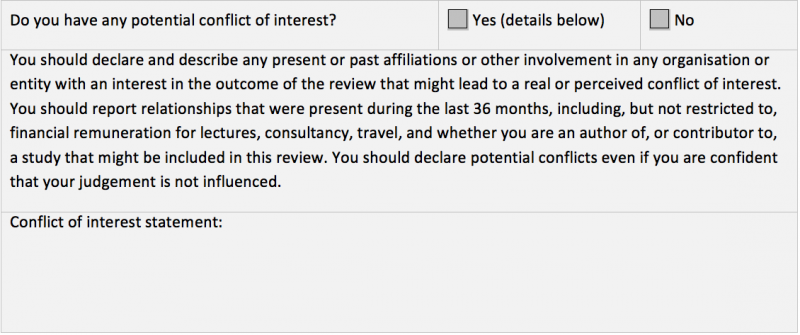
Peer reviewers must declare any potential conflicts of interest every time they undertake peer review of a Cochrane Review.
Standard Cochrane peer review checklists include a declaration of potential conflicts of interest (see Figure). If a Cochrane Review Group (CRG) uses a modified version of the checklist, it must include this question, without any amendments. CRGs that do not use the standard checklists, and the Diagnostic Test Accuracy (DTA) Editorial Team, must ensure that this question is included in the materials sent to the peer reviewer. The response from the peer reviewer should be reviewed and recorded in Archie.

Peer reviewers must state their current affiliations(s) clearly. People with a direct financial interest in a particular intervention should not peer review a Cochrane Review of that intervention. If a peer reviewer is an author of an included/excluded trial or study (study author) this should be made clear in the Conflict of Interest statement, but they can remain a peer reviewer, as long as there is no financial interest in the intervention.
It is the responsibility of the CRG or DTA Editorial Team to decide whether potential conflicting interests are sufficient to withdraw the participation of the peer reviewer and to invite an alternative peer reviewer. If the CRG or DTA Editorial Team considers the conflict of interest to be minor and agrees that it is unlikely to affect the judgement of the peer reviewer, the CRG or DTA Editorial Team must disclose the potential conflict of interest to the review authors when sharing the peer reviewer’s comments. If the CRG or DTA Editorial Team considers the conflict of interest to be major and agrees that it is possible that it will affect the judgement of the peer reviewer, the peer reviewer must be replaced.
If necessary, the CRG or DTA Editorial Team may request advice from the Cochrane funding arbiter.