Cochrane has approved standards for the content of plain language summaries of Cochrane Reviews. These standards specify the key messages from Cochrane Reviews that should be included in the plain language summary. They include the requirement that text be written in plain English, so the core components of writing for a lay audience are presented in the rationale for the first standard.
The standards provide authors with guidance on the composition of plain language summaries. Cochrane Review Groups will be able to direct authors to these standards for use in writing plain language summaries. Editors and review authors are expected to ensure that all plain language summaries are compliant with these standards.
Each standard is given a status of either mandatory (defined as compliance required for publication) or highly desirable (defined as expected but may be justifiably not done). The standards and additional information are available at methods.cochrane.org/mecir.
The process of writing plain language summaries: drafting, editing and final approval
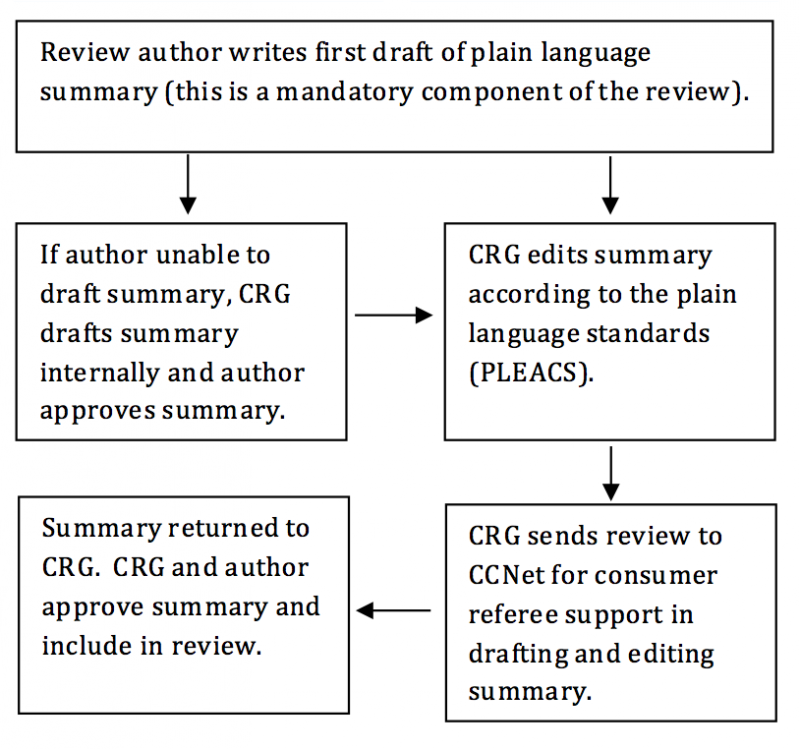
The first draft of the plain language summary should be written by the review author and submitted with the review to the relevant CRG. The writing of plain language summaries, however, is a specific skill, and review authors and CRGs may need support. Many CRGs have this skill within their editorial team, but where this is not available, a central support service will assist CRGs in writing and editing plain language summaries if they choose to access this support. The following flow chart outlines the use of this service:
The central summary support service will be maintained by the Cochrane Consumer Network (ccnet-contact@cochrane.de), with the support of the Cochrane Editorial Unit. CRGs wishing to access this service should send the review to the above address. The ownership and final approval of the plain language summary, as a mandatory part of the review, remains with the CRG and the review author.